HHS OIG: A BRIEF HISTORY
A U.S. federal Inspector General (IG) is the head of an independent, non-partisan organization established within each executive branch agency. It is assigned to audit and investigate the agency’s operations to prevent fraud waste and abuse within each agency as well as to detect and address crimes against the agency. Currently, there are 73 OIG offices within the federal government. Each office also employs criminal investigators, typically special agents to investigate internal or external criminal activity.
The Health and Human Services, Office of Inspector General (OIG) was established in 1976. With approximately 1700 professional employees and contractors, the HHS OIG is the federal government’s largest OIG office. HHS OIG was formed to protect the integrity of programs carried out by the United States Department of Health and Human Services (HHS). This was to be done by safeguarding the health and welfare of the beneficiaries of HHS OIG programs.
Each of these 1700 team members are driven by the same goal: to fight waste, fraud and abuse in Medicare, Medicaid and more than 100 other HHS programs. These programs receive some of the largest funding allocations from the federal budget. The vast sums of government funding necessary to support HHS agencies create significant risks for fraud, waste and abuse.
For fiscal year 2021, HHS OIG requested $419 million to oversee HHS programs. With these resources HHS OIG oversees more than 1.3 trillion dollars in HHS spending, which represents approximately 25% of every federal dollar spent. This includes healthcare, clinal research, epidemiology, public health and human services, and education.
Here is a brief timeline of OIG’s development:
October 15, 1976– President Ford signed a law legislation creating an Office of Inspector General (OIG) at the Department of Health, Education and Welfare (HEW). In 1980, HEW OIG became HHS OIG, after HEW was revamped as the Department of Health and Human Services (HHS).
October 27, 1986– President Reagan signed an update to the False Claims Act to combat fraud against federal government programs. Originally signed by President Lincoln in 1863 and known as the “Lincoln Law,” this Act criminalized false claims or statements against the federal government. The Reagan update also codified double damages against offenders and a $2,000 penalty for every false claim. As of June 19, 2020, each False Claims Act violation holds a minimum penalty of $11,665 and a maximum penalty of 23,331.
August 21, 1996– President Clinton signed the Health Insurance Portability and Accountability Act of 1996 (HIPAA), which paved the way to the coordination of Federal law enforcement efforts through the establishment of the Health Care Fraud and Abuse Control (HCFAC) Program.
Through these precursors, The Office of Inspector General has been able to carry out its goals and has returned approximately $50 billion to the Medicare Trust Funds since 1997.
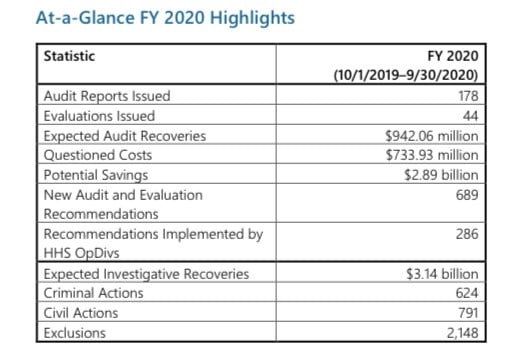
Despite the significant challenges of the COVID-19 public health emergency, in FY2020 (October 1, 2019-September 30, 2020), HHS OIG accomplished the following:
On March 19, 2021, Xavier Becerra assumed office as the 25th United States Secretary of Health and Human Services after being nominated by President Biden. It is expected that President Joe Biden will also appoint a new HHS Inspector General. The current Acting Inspector General, Christie Grimm, was appointed by former President Donald Trump on January 1, 2020.
For more information about HHS-OIG please see:
http://oig.hhs.gov/about-oig/about-us/index.asp
https://oig.hhs.gov/reports-and-publications/strategic-plan/index.asp
https://oig.hhs.gov/publications/docs/hcfac/FY2019-hcfac.pdf
https://oig.hhs.gov/documents/root/7/OIG-Strategic-Plan-2020-2025.pdf
To learn more about Exclusion Screening, visit our OFFICE OF THE INSPECTOR GENERAL OIG EXCLUSION LIST page.



































